Information
Version: B | 1.2 (2022-07-20)
Please note: This part of the profile is currently being revised.
WelfareScore | farm
Condensed assessment of the species' likelihood and potential for good fish welfare in aquaculture, based on ethological findings for 10 crucial criteria.
- Li = Likelihood that the individuals of the species experience good welfare under minimal farming conditions
- Po = Potential of the individuals of the species to experience good welfare under high-standard farming conditions
- Ce = Certainty of our findings in Likelihood and Potential
WelfareScore = Sum of criteria scoring "High" (max. 10)
General remarks
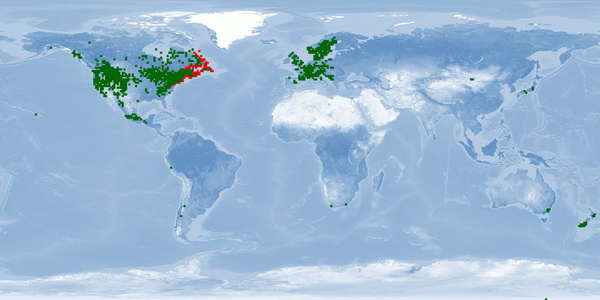
Salvelinus fontinalis is farmed not only for feeding purposes but also for recreational fishing, especially in North America. Considered invasive in several countries, where adverse ecological impact after introduction has been reported. Nevertheless, there are many biological and ethological aspects that are not respected in usual farming conditions. This species naturally swims long distances, which makes it challenging for rearing facilities to fulfill its spatial needs. Reproduction is induced through highly invasive techniques, and substrate needs are complex to assure in farms. In addition, there is a severe lack of knowledge concerning its biology, namely in aspects that are directly related to farming such as stress, malformation rates and sustainable feeds. Further research should be directed into these issues, as well as on manipulation (e.g. spawning, humane stunning and slaughtering protocols) and environmental enrichment. Selecting non-migratory strains for rearing could help to minimize harmful effects of confinement.
1 Home range
Many species traverse in a limited horizontal space (even if just for a certain period of time per year); the home range may be described as a species' understanding of its environment (i.e., its cognitive map) for the most important resources it needs access to.
What is the probability of providing the species' whole home range in captivity?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


2 Depth range
Given the availability of resources (food, shelter) or the need to avoid predators, species spend their time within a certain depth range.
What is the probability of providing the species' whole depth range in captivity?
It is unclear for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


3 Migration
Some species undergo seasonal changes of environments for different purposes (feeding, spawning, etc.), and to move there, they migrate for more or less extensive distances.
What is the probability of providing farming conditions that are compatible with the migrating or habitat-changing behaviour of the species?
It is unclear for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


4 Reproduction
A species reproduces at a certain age, season, and sex ratio and possibly involving courtship rituals.
What is the probability of the species reproducing naturally in captivity without manipulation of theses circumstances?
It is low for minimal and high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


5 Aggregation
Species differ in the way they co-exist with conspecifics or other species from being solitary to aggregating unstructured, casually roaming in shoals or closely coordinating in schools of varying densities.
What is the probability of providing farming conditions that are compatible with the aggregation behaviour of the species?
It is low for minimal and high-standard farming conditions. Our conclusion is based on a low amount of evidence.


6 Aggression
There is a range of adverse reactions in species, spanning from being relatively indifferent towards others to defending valuable resources (e.g., food, territory, mates) to actively attacking opponents.
What is the probability of the species being non-aggressive and non-territorial in captivity?
It is low for minimal and high-standard farming conditions. Our conclusion is based on a low amount of evidence.


7 Substrate
Depending on where in the water column the species lives, it differs in interacting with or relying on various substrates for feeding or covering purposes (e.g., plants, rocks and stones, sand and mud, turbidity).
What is the probability of providing the species' substrate and shelter needs in captivity?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a high amount of evidence.


8 Stress
Farming involves subjecting the species to diverse procedures (e.g., handling, air exposure, short-term confinement, short-term crowding, transport), sudden parameter changes or repeated disturbances (e.g., husbandry, size-grading).
What is the probability of the species not being stressed?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a low amount of evidence.


9 Malformations
Deformities that – in contrast to diseases – are commonly irreversible may indicate sub-optimal rearing conditions (e.g., mechanical stress during hatching and rearing, environmental factors unless mentioned in crit. 3, aquatic pollutants, nutritional deficiencies) or a general incompatibility of the species with being farmed.
What is the probability of the species being malformed rarely?
There are no findings for minimal and high-standard farming conditions.


10 Slaughter
The cornerstone for a humane treatment is that slaughter a) immediately follows stunning (i.e., while the individual is unconscious), b) happens according to a clear and reproducible set of instructions verified under farming conditions, and c) avoids pain, suffering, and distress.
What is the probability of the species being slaughtered according to a humane slaughter protocol?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


Side note: Domestication
Teletchea and Fontaine introduced 5 domestication levels illustrating how far species are from having their life cycle closed in captivity without wild input, how long they have been reared in captivity, and whether breeding programmes are in place.
What is the species’ domestication level?
DOMESTICATION LEVEL 5 41, fully domesticated.
Side note: Forage fish in the feed
450-1,000 milliard wild-caught fishes end up being processed into fish meal and fish oil each year which contributes to overfishing and represents enormous suffering. There is a broad range of feeding types within species reared in captivity.
To what degree may fish meal and fish oil based on forage fish be replaced by non-forage fishery components (e.g., poultry blood meal) or sustainable sources (e.g., soybean cake)?
All age classes: WILD: Carnivorous 42 43 26 25. FARM: No protocol available for feeding without components of forage fishery. Replacement of fish meal and fish oil not reported in literature.
Glossary
ALEVINS = larvae until the end of yolk sac absorption, for details ➝ Findings 10.1 Ontogenetic development
ANADROMOUS = migrating from the sea into fresh water to spawn
BENTHIC = living at the bottom of a body of water, able to rest on the floor
DOMESTICATION LEVEL 5 = selective breeding programmes are used focusing on specific goals 41
FARM = setting in farming environment or under conditions simulating farming environment in terms of size of facility or number of individuals
JUVENILES = fully developed but immature individuals, for details ➝ Findings 10.1 Ontogenetic development
SPAWNERS = adults during the spawning season; in farms: adults that are kept as broodstock
WILD = setting in the wild
Bibliography
2 Shelton, James L. 1994. Trout Production. Aquaculture Technical Series. Athens, Georgia: Cooperative Extension Service, The University of Georgia College of Agricultural and Environmental Sciences.
3 Gowan, C, and K D Fausch. 1996. Mobile brook trout in two high-elevation Colorado streams: reevaluating the concept of restricted movement. Canadian Journal of Fisheries and Aquatic Sciences 53: 1370–1381. https://doi.org/10.1139/f96-058.
4 Saunders, Lloyd H., and G. Power. 1970. Population Ecology of the Brook Trout, Salvelinus fontinalis, in Matamek Lake, Quebec. Journal of the Fisheries Research Board of Canada 27: 413–424. https://doi.org/10.1139/f70-051.
5 Castonguay, Martin, Gérard J. FitzGerald, and Yvon Côté. 1982. Life history and movements of anadromous brook charr, Salvelinus frontalis, in the St-Jean River, Gaspé, Quebec. Canadian Journal of Zoology 60: 3084–3091. https://doi.org/10.1139/z82-392.
6 Fischer, Gregory J., James Held, Christopher Hartleb, and Jeffrey Malison. 2009. Evaluation of brook trout production in a coldwater recycle aquaculture system. Aquacultural Engineering 41: 109–113. https://doi.org/10.1016/j.aquaeng.2009.06.012.
7 Curry, R. Allen, David Sparks, and Jacob van de Sande. 2002. Spatial and Temporal Movements of a Riverine Brook Trout Population. Transactions of the American Fisheries Society 131: 551–560. https://doi.org/10.1577/1548-8659(2002)131<0551:SATMOA>2.0.CO;2.
8 Curry, R. Allen, and David L. G. Noakes. 1995. Groundwater and the selection of spawning sites by brook trout (Salvelinus fontinalis). Canadian Journal of Fisheries and Aquatic Sciences 52: 1733–1740. https://doi.org/10.1139/f95-765.
9 Blanchfield, P J, and M S Ridgway. 1997. Reproductive timing and use of redd sites by lake-spawning brook trout (Salvelinus fontinalis). Canadian Journal of Fisheries and Aquatic Sciences 54: 747–756. https://doi.org/10.1139/f96-344.
10 Chisholm, Ian M., Wayne A. Hubert, and Thomas A. Wesche. 1987. Winter Stream Conditions and Use of Habitat by Brook Trout in High-Elevation Wyoming Streams. Transactions of the American Fisheries Society 116: 176–184. https://doi.org/10.1577/1548-8659(1987)116<176:WSCAUO>2.0.CO;2.
11 Cunjak, Richard A., and Geoffrey Power. 1986. Winter Habitat Utilization by Stream Resident Brook Trout (Salvelinus fontinalis) and Brown Trout (Salmo trutta). Canadian Journal of Fisheries and Aquatic Sciences 43: 1970–1981. https://doi.org/10.1139/f86-242.
12 Wilder, D. G. 1952. A Comparative Study of Anadromous and Freshwater Populations of Brook Trout (Salvelinus fontinalis (Mitchill)). Journal of the Fisheries Research Board of Canada 9: 169–203. https://doi.org/10.1139/f52-012.
13 McCormick, Stephen D., and Robert J. Naiman. 1984. Osmoregulation in the brook trout, Salvelinus fontinalis—II. Effects of size, age and photoperiod on seawater survival and ionic regulation. Comparative Biochemistry and Physiology Part A: Physiology 79: 17–28. https://doi.org/10.1016/0300-9629(84)90704-7.
14 Doucett, Richard R., William Hooper, and Geoff Power. 1999. Identification of Anadromous and Nonanadromous Adult Brook Trout and Their Progeny in the Tabusintac River, New Brunswick, by Means of Multiple-Stable-Isotope Analysis. Transactions of the American Fisheries Society 128: 278–288. https://doi.org/10.1577/1548-8659(1999)128<0278:IOAANA>2.0.CO;2.
15 Morinville, Geneviève R, and Joseph B Rasmussen. 2003. Early juvenile bioenergetic differences between anadromous and resident brook trout (Salvelinus fontinalis). Canadian Journal of Fisheries and Aquatic Sciences 60: 401–410. https://doi.org/10.1139/f03-036.
16 Crespel, A., A. Dupont-Prinet, L. Bernatchez, G. Claireaux, R. Tremblay, and C. Audet. 2017. Divergence in physiological factors affecting swimming performance between anadromous and resident populations of brook charr Salvelinus fontinalis. Journal of Fish Biology 90: 2170–2193. https://doi.org/10.1111/jfb.13300.
17 Warren, Dana R., Jason M. Robinson, Daniel C. Josephson, Daniel R. Sheldon, and Clifford E. Kraft. 2012. Elevated summer temperatures delay spawning and reduce redd construction for resident brook trout (Salvelinus fontinalis). Global Change Biology 18: 1804–1811. https://doi.org/10.1111/j.1365-2486.2012.02670.x.
18 Snucins, E. J., R. Allen Curry, and J. M. Gunn. 1992. Brook trout (Salvelinus fontinalis) embryo habitat and timing of alevin emergence in a lake and a stream. Canadian Journal of Zoology 70: 423–427. https://doi.org/10.1139/z92-064.
19 Mirza, Reehan S., Douglas P. Chivers, and Jean-Guy J. Godin. 2001. Brook charr alevins alter timing of nest emergence in response to chemical cues from fish predators. Journal of Chemical Ecology 27: 1775–1785. https://doi.org/10.1023/A:1010404624556.
20 Galbreath, Peter F., and Barbara L. Samples. 2000. Optimization of Thermal Shock Protocols for Induction of Triploidy in Brook Trout. North American Journal of Aquaculture 62: 249–259. https://doi.org/10.1577/1548-8454(2000)062<0249:OOTSPF>2.0.CO;2.
21 Cooney, K. 2014. Longitudinal Differences in Brook Trout Density and Mean Length in Headwater Streams of Western Massachusetts. Amherst, Massachussets: University of Massachussets, Amherst.
22 MacMillan, J.L., D. Caissie, T.J. Marshall, and L. Hinks. 2008. Population indices of brook trout (Salvelinus fontinalis), Atlantic salmon (Salmo salar), and salmonid competitors in relation to summer water temperature and habitat parameters in 100 streams in Nova Scotia. Canadian Technical Report of Fisheries and Aquatic Sciences 2819. Moncton, NB: Department of Fisheries and Oceans.
23 Wagner, Tyler, Jefferson T. Deweber, Jason Detar, David Kristine, and John A. Sweka. 2014. Spatial and Temporal Dynamics in Brook Trout Density: Implications for Population Monitoring. North American Journal of Fisheries Management 34: 258–269. https://doi.org/10.1080/02755947.2013.847878.
24 Ridgway, M S, and P J Blanchfield. 1998. Borrok trout spawning areas in lakes. Ecology of Freshwater Fish 7: 140–145.
25 Williams, D. Dudley. 1981. The First Diets of Postemergent Brook Trout (Salvelinus fontinalis) and Atlantic Salmon (Salmo salar) Alevins in a Quebec River. Canadian Journal of Fisheries and Aquatic Sciences 38: 765–771. https://doi.org/10.1139/f81-104.
26 Grant, James W. A. 1990. Aggressiveness and the Foraging Behaviour of Young-of-the-Year Brook Charr (Salvelinus fontinalis). Canadian Journal of Fisheries and Aquatic Sciences 47: 915–920. https://doi.org/10.1139/f90-105.
27 Moyle, Peter B. 1969. Comparative Behavior of Young Brook Trout of Domestic and Wild Origin. The Progressive Fish-Culturist 31: 51–56. https://doi.org/10.1577/1548-8640(1969)31[51:CBOYBT]2.0.CO;2.
28 NOT FOUND
29 Blanchfield, Paul J., and Mark S. Ridgway. 1999. The cost of peripheral males in a brook trout mating system. Animal Behaviour 57: 537–544. https://doi.org/10.1006/anbe.1998.1014.
30 Grant, James W. A., and David L. G. Noakes. 1987. Escape Behaviour and Use of Cover by Young-of-the-Year Brook Trout, Salvelinus fontinalis. Canadian Journal of Fisheries and Aquatic Sciences 44: 1390–1396. https://doi.org/10.1139/f87-167.
31 NOT FOUND
32 Webster, Dwight A. 1962. Artificial Spawning Facilities for Brook Trout, Salvelinus fontinalis. Transactions of the American Fisheries Society 91: 168–174. https://doi.org/10.1577/1548-8659(1962)91[168:ASFFBT]2.0.CO;2.
33 Curry, R. Allen, David L. G. Noakes, and George E. Morgan. 1995. Groundwater and the incubation and emergence of brook trout (Salvelinus fontinalis). Canadian Journal of Fisheries and Aquatic Sciences 52: 1741–1749. https://doi.org/10.1139/f95-766.
34 Biron, Michel, and Tillmann J. Benfey. 1994. Cortisol, glucose and hematocrit changes during acute stress, cohort sampling, and the diel cycle in diploid and triploid brook trout (Salvelinus fontinalis Mitchill). Fish Physiology and Biochemistry 13: 153–160. https://doi.org/10.1007/BF00004340.
35 Barton, Bruce A. 2000. Salmonid Fishes Differ in Their Cortisol and Glucose Responses to Handling and Transport Stress. North American Journal of Aquaculture 62: 12–18. https://doi.org/10.1577/1548-8454(2000)062<0012:SFDITC>2.0.CO;2.
36 Lund, Susan G, Mervyn E.A Lund, and Bruce L Tufts. 2003. Red blood cell Hsp 70 mRNA and protein as bio-indicators of temperature stress in the brook trout (Salvelinus fontinalis). Canadian Journal of Fisheries and Aquatic Sciences 60: 460–470. https://doi.org/10.1139/f03-039.
37 McDonald, D. G., M. D. Goldstein, and C. Mitton. 1993. Responses of Hatchery-Reared Brook Trout, Lake Trout, and Splake to Transport Stress. Transactions of the American Fisheries Society 122: 1127–1138. https://doi.org/10.1577/1548-8659(1993)122<1127:ROHRBT>2.3.CO;2.
38 Fairgrieve, W. 2009. Cultured Aquatic Species Information Programme. Oncorhynchus kisutch. Rome: FAO Fisheries and Aquaculture Department.
39 LocalCoho Farms. 2021. Personal communication.
40 Lines, J. A., D. H. Robb, S. C. Kestin, S. C. Crook, and T. Benson. 2003. Electric stunning: a humane slaughter method for trout. Aquacultural Engineering 28: 141–154. https://doi.org/10.1016/S0144-8609(03)00021-9.
41 Teletchea, Fabrice, and Pascal Fontaine. 2012. Levels of domestication in fish: implications for the sustainable future of aquaculture. Fish and Fisheries 15: 181–195. https://doi.org/10.1111/faf.12006.
42 Leonard, Justin W. 1942. Some Observations on the Winter Feeding Habits of Brook Trout Fingerlings in Relation to Natural Food Organisms Present. Transactions of the American Fisheries Society 71: 219–227. https://doi.org/10.1577/1548-8659(1941)71[219:SOOTWF]2.0.CO;2.
43 Allan, J. David. 1981. Determinants of Diet of Brook Trout (Salvelinus fontinalis) in a Mountain Stream. Canadian Journal of Fisheries and Aquatic Sciences 38: 184–192. https://doi.org/10.1139/f81-024.