Information
Version: B | 1.4 (2022-07-29)
Please note: This part of the profile is currently being revised.
WelfareScore | farm
Condensed assessment of the species' likelihood and potential for good fish welfare in aquaculture, based on ethological findings for 10 crucial criteria.
- Li = Likelihood that the individuals of the species experience good welfare under minimal farming conditions
- Po = Potential of the individuals of the species to experience good welfare under high-standard farming conditions
- Ce = Certainty of our findings in Likelihood and Potential
WelfareScore = Sum of criteria scoring "High" (max. 10)
General remarks
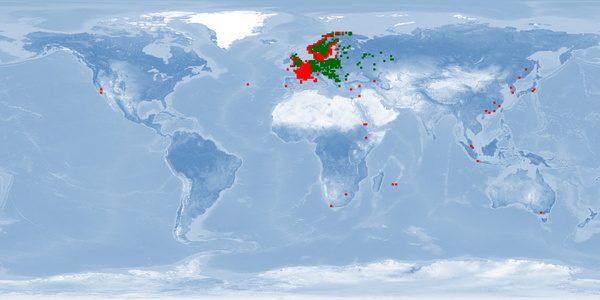
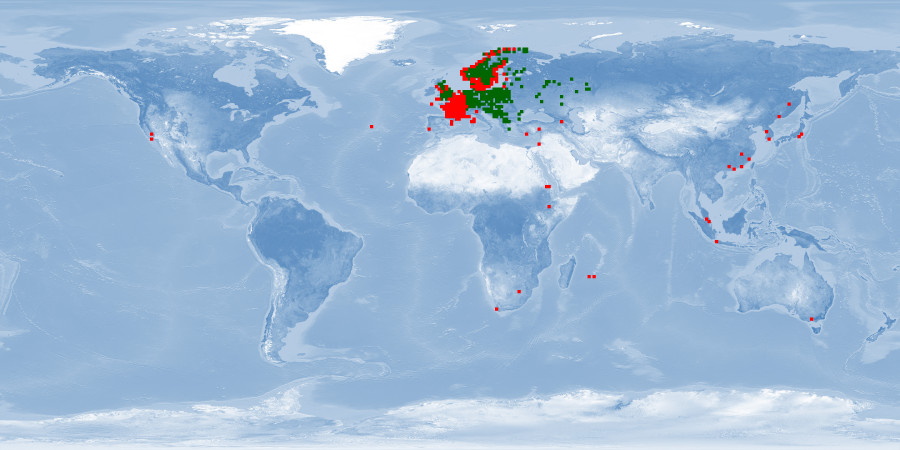
Carassius carassius is a freshwater fish that inhabits ponds, lakes, rivers, and reservoirs in several countries in Asia and Europe. This carp appears in IUCN Red List with a substantial decline in most of its native range in Europe. It is found in rich vegetation in slow-moving waters with sand and gravel and can tolerate a wide range of environmental conditions – including anoxia – but usually does not occur in waters with rich ichthyofauna. This carp, which migrates in fresh water to spawn when the temperature is warmer, is considered a relatively inactive fish. Aquaculture of C. carassius was limited to China and Japan until the 1960s and then gradually expanded to many other countries. It is considered an omnivore fish with relatively slow growth that explores the water column, especially dwelling in the bottom layer, thus being difficult to harvest. Despite that, it occupies a high position among all cultured freshwater fishes worldwide, being reared also for conservation programmes. C. carassius is frequently cultured in earthen ponds, also in pens and rice paddies. Polycultures with other carps is common. This fish is usually sold live or fresh in local markets, and this can happen before it reaches maturity. Further studies about wild information, especially about migration, aggregation, and aggression of this species, are needed. Considering farming conditions, the need for future research focused on depth range, substrate availability, stress response, and stunning and slaughter processes is highlighted. Moreover, as most available information about farms are from sources focused on conservation purposes, more research about production conditions are still needed.
1 Home range
Many species traverse in a limited horizontal space (even if just for a certain period of time per year); the home range may be described as a species' understanding of its environment (i.e., its cognitive map) for the most important resources it needs access to.
What is the probability of providing the species' whole home range in captivity?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a low amount of evidence.


2 Depth range
Given the availability of resources (food, shelter) or the need to avoid predators, species spend their time within a certain depth range.
What is the probability of providing the species' whole depth range in captivity?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a low amount of evidence.


3 Migration
Some species undergo seasonal changes of environments for different purposes (feeding, spawning, etc.), and to move there, they migrate for more or less extensive distances.
What is the probability of providing farming conditions that are compatible with the migrating or habitat-changing behaviour of the species?
It is low for minimal farming conditions. It is high for high-standard farming conditions. Our conclusion is based on a low amount of evidence.


4 Reproduction
A species reproduces at a certain age, season, and sex ratio and possibly involving courtship rituals.
What is the probability of the species reproducing naturally in captivity without manipulation of theses circumstances?
It is low for minimal and high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


5 Aggregation
Species differ in the way they co-exist with conspecifics or other species from being solitary to aggregating unstructured, casually roaming in shoals or closely coordinating in schools of varying densities.
What is the probability of providing farming conditions that are compatible with the aggregation behaviour of the species?
It is unclear for minimal and high-standard farming conditions. Our conclusion is based on a low amount of evidence.


6 Aggression
There is a range of adverse reactions in species, spanning from being relatively indifferent towards others to defending valuable resources (e.g., food, territory, mates) to actively attacking opponents.
What is the probability of the species being non-aggressive and non-territorial in captivity?
It is low for minimal and high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


7 Substrate
Depending on where in the water column the species lives, it differs in interacting with or relying on various substrates for feeding or covering purposes (e.g., plants, rocks and stones, sand and mud, turbidity).
What is the probability of providing the species' substrate and shelter needs in captivity?
It is low for minimal farming conditions. It is high for high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


8 Stress
Farming involves subjecting the species to diverse procedures (e.g., handling, air exposure, short-term confinement, short-term crowding, transport), sudden parameter changes or repeated disturbances (e.g., husbandry, size-grading).
What is the probability of the species not being stressed?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a low amount of evidence.


9 Malformations
Deformities that – in contrast to diseases – are commonly irreversible may indicate sub-optimal rearing conditions (e.g., mechanical stress during hatching and rearing, environmental factors unless mentioned in crit. 3, aquatic pollutants, nutritional deficiencies) or a general incompatibility of the species with being farmed.
What is the probability of the species being malformed rarely?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a low amount of evidence.


10 Slaughter
The cornerstone for a humane treatment is that slaughter a) immediately follows stunning (i.e., while the individual is unconscious), b) happens according to a clear and reproducible set of instructions verified under farming conditions, and c) avoids pain, suffering, and distress.
What is the probability of the species being slaughtered according to a humane slaughter protocol?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a low amount of evidence.


Side note: Domestication
Teletchea and Fontaine introduced 5 domestication levels illustrating how far species are from having their life cycle closed in captivity without wild input, how long they have been reared in captivity, and whether breeding programmes are in place.
What is the species’ domestication level?
DOMESTICATION LEVEL 5 36, fully domesticated.
Side note: Forage fish in the feed
450-1,000 milliard wild-caught fishes end up being processed into fish meal and fish oil each year which contributes to overfishing and represents enormous suffering. There is a broad range of feeding types within species reared in captivity.
To what degree may fish meal and fish oil based on forage fish be replaced by non-forage fishery components (e.g., poultry blood meal) or sustainable sources (e.g., soybean cake)?
All age classes: WILD: omnivorous 6 17 14, LARVAE and FRY feeding on zooplankton 6 3, older age classes on plankton, BENTHIC invertebrates 6 3 14, plant materials, and detritus 6 14. FARM: feed usually consists of by-products from grain processing and oil extraction with <10% fish meal, but no fish meal is necessary when C. carassius is kept as a secondary species in polycultures 6.
Glossary
BENTHIC = living at the bottom of a body of water, able to rest on the floor
DOMESTICATION LEVEL 5 = selective breeding programmes are used focusing on specific goals 36
FARM = setting in farming environment or under conditions simulating farming environment in terms of size of facility or number of individuals
FISHES = Using "fishes" instead of "fish" for more than one individual - whether of the same species or not - is inspired by Jonathan Balcombe who proposed this usage in his book "What a fish knows". By referring to a group as "fishes", we acknowledge the individuals with their personalities and needs instead of an anonymous mass of "fish".
FRY = larvae from external feeding on, for details ➝ Findings 10.1 Ontogenetic development
IND = individuals
JUVENILES = fully developed but immature individuals, for details ➝ Findings 10.1 Ontogenetic development
LAB = setting in laboratory environment
LARVAE = hatching to mouth opening, for details ➝ Findings 10.1 Ontogenetic development
PHOTOPERIOD = duration of daylight
POTAMODROMOUS = migrating within fresh water
SPAWNERS = adults during the spawning season; in farms: adults that are kept as broodstock
WILD = setting in the wild
Bibliography
2 Vinterstare, J., K. Hulthén, P. A. Nilsson, H. Nilsson Sköld, and C. Brönmark. 2020. Experimental manipulation of perceived predation risk and cortisol generates contrasting trait trajectories in plastic crucian carp. Journal of Experimental Biology 223: jeb213611. https://doi.org/10.1242/jeb.213611.
3 Penttinen, O. P., and I. J. Holopainen. 1992. Seasonal feeding activity and ontogenetic dietary shifts in crucian carp, Carassius carassius. Environmental Biology of Fishes 33: 215–221. https://doi.org/10.1007/BF00002566.
4 Radtke, G. 2011. Comparison of seasonal activity of the lake minnow, (Pall.), and crucian carp, (L.), in small water bodies in northern Poland. Fisheries & Aquatic Life 19: 175–182. https://doi.org/10.2478/v10086-011-0022-7.
5 Żarski, D., K. Targońska, S. Krejszeff, M. Kwiatkowski, K. Kupren, and D. Kucharczyk. 2011. Influence of stocking density and type of feed on the rearing of crucian carp, Carassius carassius (L.), larvae under controlled conditions. Aquaculture International 19: 1105–1117. https://doi.org/10.1007/s10499-011-9427-y.
6 Weimin, M. Cultured Aquatic Species Information Programme. Carassius carassius. Rome: FAO Fisheries and Aquaculture Department.
7 Tonn, W. M., C. A. Paszkowski, and I. J. Holopainen. 1989. Responses of crucian carp populations to differential predation pressure in a manipulated pond. Canadian Journal of Zoology 67: 2841–2849. https://doi.org/10.1139/z89-400.
8 Meo, I., K. Østbye, K. K. Kahilainen, B. Hayden, C. H. Fagertun, and A. B. S. Poléo. 2021. Predator community and resource use jointly modulate the inducible defense response in body height of crucian carp. Ecology and Evolution 11: 2072–2085. https://doi.org/10.1002/ece3.7176.
9 Zargar, U. R., M. Z. Chishti, A. R. Yousuf, and A. Fayaz. 2012. Infection level of monogenean gill parasite, Diplozoon kashmirensis (Monogenea, Polyopisthocotylea) in the Crucian Carp, Carassius carassius from lake ecosystems of an altered water quality: What factors do have an impact on the Diplozoon infection? Veterinary Parasitology 189: 218–226. https://doi.org/10.1016/j.vetpar.2012.04.029.
10 Zargar, U. R., A. R. Yousuf, Basharat Mushtaq, and Dilafroza Jan. 2012. Length-Weight Relationship of the Crucian carp, Carassius carassius in Relation to Water Quality, Sex and Season in Some Lentic Water Bodies of Kashmir Himalayas. Turkish Journal of Fisheries and Aquatic Sciences 12: 683–689.
11 Ahn, C. H., S. Lee, H. M. Song, K.-W. Bang, J. Park, and J. C. Joo. 2016. Evaluation of the performance for an artificial deep pool (ADP) as an alternative fish shelter. Ecological Engineering 93: 37–45. https://doi.org/10.1016/j.ecoleng.2016.05.008.
12 Kapusta, A., K. Partyka, B. Szczepkowska, S. Jarmołowicz, M. Hopko, I. Piotrowska, A. Kowalska, and Z. Zakęś. 2013. Impact of diet and culture conditions on the body shape of crucian carp (Carassius carassius L.). Journal of Applied Animal Research 41: 462–469. https://doi.org/10.1080/09712119.2013.792738.
13 Targońska, K., D. Żarski, T. Müller, S. Krejszeff, K. Kozłowski, F. Demény, B. Urbányi, and D. Kucharczyk. 2012. Controlled reproduction of the crucian carp Carassius carassius (L.) combining temperature and hormonal treatment in spawners. Journal of Applied Ichthyology 28: 894–899. https://doi.org/10.1111/jai.12073.
14 Froese, R., and D. Pauly. 2021. Carassius carassius, Crucian carp: fisheries, aquaculture. World Wide Web electronic publication. www.fishbase.org.
15 Allardi, J., and P. Keith. 1991. Atlas preliminaire des poissons d eau douce de France. Coll. Patrimoines Naturels, vol. 4.
16 Riede, K. 2004. Global register of migratory species - from global to regional scales. Final report of the R&D Projekt 808 05 081. Bonn, Germany: Federal Agency for Nature Conservation.
17 Zha, W., M. Huang, and Y. Zeng. 2019. Swimming behavior of crucian carp in an open channel with sudden expansion. River Research and Applications 35: 1499–1510. https://doi.org/10.1002/rra.3500.
18 Demény F., Müllerné Trenovszki M., Sokoray S. V., Hegyi Á., Urbányi B., Żarski D., Ács B., Miljanovic B., Specziár A., and Müller T. 2012. Relative efficiencies of Artemia nauplii, dry food and mixed food diets in intensive rearing of larval Crucian carp (Carassius carassius L.). Turkish journal of Fisheries and aquatic Sciences 12: 691–698.
19 Busst, G. M. A., and J. R. Britton. 2015. Quantifying the growth consequences for crucian carp Carassius carassius of competition from non-native fishes. Ecology of Freshwater Fish 24: 489–492. https://doi.org/10.1111/eff.12155.
20 Sikorska, J., E. Kondera, R. Kamiński, K. Ługowska, M. Witeska, and J. Wolnicki. 2018. Effect of four rearing water temperatures on some performance parameters of larval and juvenile crucian carp, Carassius carassius, under controlled conditions. Aquaculture Research 49: 3874–3880. https://doi.org/10.1111/are.13855.
21 Ruchin, A. B. 2019. Effect of illumination on growth and behavior of two carp fish species (Carassius gibelio and C. carassius). Periódico Tchê Química 16: 8–17.
22 Cejko, B. I., and D. Kucharczyk. 2015. Application of dopaminergic antagonist: Metoclopramide, in reproduction of crucian carp Carassius carassius (L.) under controlled conditions. Animal Reproduction Science 160: 74–81. https://doi.org/10.1016/j.anireprosci.2015.07.005.
23 Wellby, I., A. Girdler, and R. Welcomme. 2010. Fisheries management: A manual for still‐water coarse fisheries. Oxford, UK: John Wiley & Sons.
24 Aho, Jussi, and Ismo J. Holopainen. 2000. Batch spawning of crucian carp (Carassius carassius (L.)) in mono- and multispecies communities. Annales Zoologici Fennici 37: 101–111.
25 Dadebo, Elias, and Daba Tugie. 2009. Some aspects of reproductive biology of the crucian carp Carassius carassius (L., 1758) (Pisces: Cyprinidae) in Lake Ziway, Ethiopia. Ethiopian Journal of Biological Sciences 8: 109–121.
26 Ali, M. S., M. G. S. Riar, M. F. Siddique, and Y. Mahmud. 2015. Effect of pituitary gland doses on induced breeding of Crucian Carp, Carassius carassius. Journal of the Bangladesh Agricultural University 13: 137–144. https://doi.org/10.22004/ag.econ.211231.
27 Holopainen, I. J., W. M. Tonn, and C. A. Paszkowski. 1997. Tales of two fish: the dichotomous biology of crucian carp (Carassius carassius (L.)) in northern Europe. Annales Zoologici Fennici 34: 1–22.
28 Piironen, Jorma, and Ismo J. Holopainen. 1988. Length structure and reproductive potential of crucian carp (Carassius carassius (L.)) populations in some small forest ponds. Annales Zoologici Fennici 25: 203–208.
29 Paszkowski, C. A., W. M. Tonn, J. Piironen, and I. J. Holopainen. 1990. Behavioral and population-level aspects of intraspecific competition in crucian carp. Annales Zoologici Fennici 27: 77–85.
30 Kottelat, Maurice, and Jörg Freyhof. 2007. Handbook of European freshwater fishes. Publications Kottelat.
31 Kasprzak, R., T. Ostaszewska, and B. Wagner. 2019. The effect of feeding commercial diets on the development of juvenile crucian carp (Carassius carassius, L.). Part 1: Skeletal deformations. Aquaculture Nutrition 25: 78–87. https://doi.org/10.1111/anu.12831.
32 Kasprzak, R., T. Ostaszewska, and M. Kamaszewski. 2019. Effects of feeding commercial diets on the development of juvenile crucian carp Carassius carassius: digestive tract abnormalities. Aquatic Biology 28: 159–173. https://doi.org/10.3354/ab00717.
33 Myszkowski, L., R. Kamiński, M. Quiros, L. A. Stanny, and J. Wolnicki. 2012. Dry diet-influenced growth, size variability, condition and body deformities in juvenile crucian carp Carassius carassius L. reared under controlled conditions. Fisheries & Aquatic Life 20: 157–163. https://doi.org/10.2478/v10086-012-0019-x.
34 Retter, Karina, Karl-Heinz Esser, Matthias Lüpke, John Hellmann, Dieter Steinhagen, and Verena Jung-Schroers. 2018. Stunning of common carp: Results from a field and a laboratory study. BMC Veterinary Research 14: 1–11. https://doi.org/10.1186/s12917-018-1530-0.
35 Rahmanifarah, K., B. Shabanpour, and A. Sattari. 2011. Effects of Clove Oil on Behavior and Flesh Quality of Common Carp (Cyprinus carpio L.) in Comparison with Pre-slaughter CO2 Stunning, Chilling and Asphyxia. Turkish Journal of Fisheries and Aquatic Sciences 11: 139–147.
36 Teletchea, Fabrice, and Pascal Fontaine. 2012. Levels of domestication in fish: implications for the sustainable future of aquaculture. Fish and Fisheries 15: 181–195. https://doi.org/10.1111/faf.12006.