Information
Version: B | 1.1 (2022-07-20)
WelfareScore | farm
Condensed assessment of the species' likelihood and potential for good fish welfare in aquaculture, based on ethological findings for 10 crucial criteria.
- Li = Likelihood that the individuals of the species experience good welfare under minimal farming conditions
- Po = Potential of the individuals of the species to experience good welfare under high-standard farming conditions
- Ce = Certainty of our findings in Likelihood and Potential
WelfareScore = Sum of criteria scoring "High" (max. 10)
General remarks
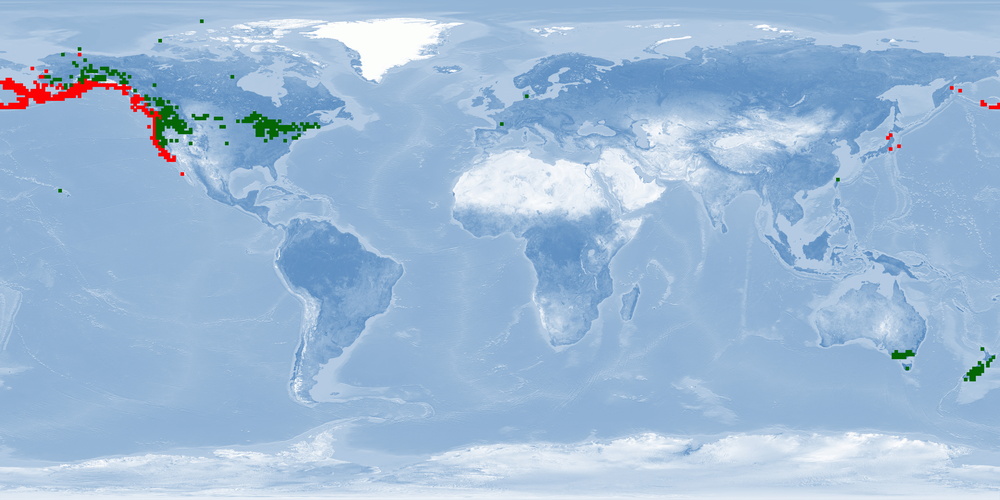
Oncorhynchus tshawytscha is a Pacific salmon species distributed from northern Hokkaido to the Anadyr River on the Asian coast and from central California to Kotzebue Sound, Alaska, on the North American coast. Two morphotypes have been described, a "spring/stream" type that remains in the streams for a year and an "fall/ocean" type that migrates to the ocean a few weeks after hatching. O. tshawytscha is anadromous: eggs hatch in streams, juveniles (parr) live in streams for one or two years or a few weeks before migrating to the ocean. In the ocean, smolts grow into adults and either stay at the coast or migrate mostly up north. When they are close to maturity, they migrate back to their original streams to spawn in the autumn, independently of when they enter the stream. Females create several nests in a defended area called redd. O. tshawytscha dies after reproduction. Triploid breeds can be used to avoid the reproductive stage in farms. Because of their need to migrate as adults, it is unlikely that current farms can provide this welfare need. Further research needs to be done to accommodate this need into farming conditions and on living offshore (home range, aggregation, aggression, substrate). O. tshawytscha was successfully transplanted in New Zealand in the late 1800s, and nowadays New Zealand is the major exporting country of O. tshawytscha. Some populations in the USA are listed as endangered or threatened.
1 Home range
Many species traverse in a limited horizontal space (even if just for a certain period of time per year); the home range may be described as a species' understanding of its environment (i.e., its cognitive map) for the most important resources it needs access to.
What is the probability of providing the species' whole home range in captivity?
It is unclear for minimal and high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


2 Depth range
Given the availability of resources (food, shelter) or the need to avoid predators, species spend their time within a certain depth range.
What is the probability of providing the species' whole depth range in captivity?
It is unclear for minimal and high-standard farming conditions. Our conclusion is based on a low amount of evidence.


3 Migration
Some species undergo seasonal changes of environments for different purposes (feeding, spawning, etc.), and to move there, they migrate for more or less extensive distances.
What is the probability of providing farming conditions that are compatible with the migrating or habitat-changing behaviour of the species?
It is low for minimal and high-standard farming conditions. Our conclusion is based on a high amount of evidence.


4 Reproduction
A species reproduces at a certain age, season, and sex ratio and possibly involving courtship rituals.
What is the probability of the species reproducing naturally in captivity without manipulation of theses circumstances?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a high amount of evidence.


5 Aggregation
Species differ in the way they co-exist with conspecifics or other species from being solitary to aggregating unstructured, casually roaming in shoals or closely coordinating in schools of varying densities.
What is the probability of providing farming conditions that are compatible with the aggregation behaviour of the species?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a low amount of evidence.


6 Aggression
There is a range of adverse reactions in species, spanning from being relatively indifferent towards others to defending valuable resources (e.g., food, territory, mates) to actively attacking opponents.
What is the probability of the species being non-aggressive and non-territorial in captivity?
It is unclear for minimal and high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


7 Substrate
Depending on where in the water column the species lives, it differs in interacting with or relying on various substrates for feeding or covering purposes (e.g., plants, rocks and stones, sand and mud, turbidity).
What is the probability of providing the species' substrate and shelter needs in captivity?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


8 Stress
Farming involves subjecting the species to diverse procedures (e.g., handling, air exposure, short-term confinement, short-term crowding, transport), sudden parameter changes or repeated disturbances (e.g., husbandry, size-grading).
What is the probability of the species not being stressed?
It is unclear for minimal and high-standard farming conditions. Our conclusion is based on a low amount of evidence.


9 Malformations
Deformities that – in contrast to diseases – are commonly irreversible may indicate sub-optimal rearing conditions (e.g., mechanical stress during hatching and rearing, environmental factors unless mentioned in crit. 3, aquatic pollutants, nutritional deficiencies) or a general incompatibility of the species with being farmed.
What is the probability of the species being malformed rarely?
It is low for minimal and high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


10 Slaughter
The cornerstone for a humane treatment is that slaughter a) immediately follows stunning (i.e., while the individual is unconscious), b) happens according to a clear and reproducible set of instructions verified under farming conditions, and c) avoids pain, suffering, and distress.
What is the probability of the species being slaughtered according to a humane slaughter protocol?
It is high for minimal and high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


Side note: Domestication
Teletchea and Fontaine introduced 5 domestication levels illustrating how far species are from having their life cycle closed in captivity without wild input, how long they have been reared in captivity, and whether breeding programmes are in place.
What is the species’ domestication level?
DOMESTICATION LEVEL 5 53, fully domesticated.
Side note: Forage fish in the feed
450-1,000 milliard wild-caught fishes end up being processed into fish meal and fish oil each year which contributes to overfishing and represents enormous suffering. There is a broad range of feeding types within species reared in captivity.
To what degree may fish meal and fish oil based on forage fish be replaced by non-forage fishery components (e.g., poultry blood meal) or sustainable sources (e.g., soybean cake)?
All age classes: WILD: carnivorous 15 20. FARM: fish meal may be partly* replaced by sustainable 54 55 or non-forage fishery components 56 or mostly* replaced by non-forage fishery components 57, and fish oil may be mostly* 58 to completely* replaced by sustainable sources 59 60, but no data found yet for ADULTS and SPAWNERS. Inclusion of soybean meal may lead to intestinal inflammation 61.
* partly = <51% – mostly = 51-99% – completely = 100%
Glossary
ALEVINS = larvae until the end of yolk sac absorption, for details ➝ Findings 10.1 Ontogenetic development
ANADROMOUS = migrating from the sea into fresh water to spawn
DOMESTICATION LEVEL 5 = selective breeding programmes are used focusing on specific goals 53
EURYHALINE = tolerant of a wide range of salinities
FARM = setting in farming environment or under conditions simulating farming environment in terms of size of facility or number of individuals
FRY = larvae from external feeding on, for details ➝ Findings 10.1 Ontogenetic development
GRILSE = adults returning from sea to home river to spawn, for details ➝ Findings 10.1 Ontogenetic development
IND = individuals
LAB = setting in laboratory environment
PARR = juvenile stage in rivers, for details ➝ Findings 10.1 Ontogenetic development
PHOTOPERIOD = duration of daylight
SMOLTS = juvenile stage migrating to the sea, for details ➝ Findings 10.1 Ontogenetic development
SPAWNERS = adults during the spawning season; in farms: adults that are kept as broodstock
WILD = setting in the wild
null = setting in farm environment
Bibliography
2 NIWA. 2020. Chinook salmon. Https://niwa.co.nz/aquaculture/species/chinook-salmon. NIWA.
3 Creative Salmon. 2021. Our Process. Creative Salmon.
4 Lovett, B. A., E. C. Firth, I. D. Tuck, J. E. Symonds, S. P. Walker, M. R. Perrott, P. S. Davie, J. S. Munday, M. A. Preece, and N. A. Herbert. 2020. Radiographic characterisation of spinal curvature development in farmed New Zealand Chinook salmon Oncorhynchus tshawytscha throughout seawater production. Scientific Reports 10. https://doi.org/10.1038/s41598-020-77121-y.
5 Perrott, M. R., J. E. Symonds, S. P. Walker, F. S. Hely, B. Wybourne, M. A. Preece, and P. S. Davie. 2018. Spinal curvatures and onset of vertebral deformities in farmed Chinook salmon, Oncorhynchus tshawytscha (Walbaum, 1792) in New Zealand. Journal of Applied Ichthyology 34: 501–511. https://doi.org/https://doi.org/10.1111/jai.13663.
6 Davie, Peter S., Seamus P. Walker, Matthew R. Perrott, Jane E. Symonds, Mark Preece, Bailey A. Lovett, and John S. Munday. 2019. Vertebral fusions in farmed Chinook salmon (Oncorhynchus tshawytscha) in New Zealand. Journal of Fish Diseases 42: 965–974. https://doi.org/https://doi.org/10.1111/jfd.13013.
7 Johnson, W S. 1984. Photoperiod Induced Delayed Maturation Of Freshwater Reared Chinook Salmon. Aquaculture 43: 279–287.
8 Chapman, D W, D E Weitkamp, T L Welsh, M B Dell, and T H Schadt. 1986. Effects of River Flow on the Distribution of Chinook Salmon Redds: 13.
9 Chapman, Wilbert McLeod. 1943. The Spawning of Chinook Salmon in the Main Columbia River. Copeia 1943: 168. https://doi.org/10.2307/1438610.
10 NOT FOUND
11 Briggs, John C. 1953. The behavior and reproduction of salmonid fishes in a small coastal stream. State of California, Department of Fish and Game, Marine Fisheries Branch.
12 Hunter, George A., Edward M. Donaldson, Eldon T. Stone, and Helen M. Dye. 1978. Induced ovulation of female chinook salmon (Oncorhynchus tshawytscha) at a production hatchery. Aquaculture 15: 99–112. https://doi.org/10.1016/0044-8486(78)90056-X.
13 Everest, F H, and D W Chapman. 1972. Habitat Selection and Spatial Interaction by Juvenile Chinook Salmon and Steelhead Trout in Two Idaho Streams. Journal of the Fisheries Research Board of Canada 29: 91–100. https://doi.org/doi:10.1139/f72-012.
14 Argue, Alexander W. 1970. Study of factors affecting exploitation of Pacific salmon in the Canadian gantlet fishery of Juan de Fuca Strait. University of British Columbia. https://doi.org/10.14288/1.0102106.
15 Healey, M.C. 1991. Life History of Chinook Salmon. In Pacific salmon life histories, 311–394. Vancouver: UBC Press.
16 Wagner, H. H., F. P. Conte, and J. L. Fessler. 1969. Development of osmotic and ionic regulation in two races of chinook salmon Oncorhynchus tshawytscha. Comparative Biochemistry and Physiology 29: 325–341.
17 Healey, M C. 1980. Utilization Of The Nanaimo River Estuary by Juvenile Chinook Salmon, Oncorhynchus Tshawytscha: 17.
18 Van Hyning, Jack M. 1951. The ocean salmon troll fishery of Oregon.
19 Gilbert, Charles Henry. 1913. Age at Maturity of the Pacific Coast Salmon of the Genus Oncorhynchus. U.S. Government Printing Office.
20 Healey, M.C. 1982. Juvenile Pacific Salmon In Estuaries: The Life Support System. In Estuarine Comparisons, 315–341. Elsevier. https://doi.org/10.1016/B978-0-12-404070-0.50025-9.
21 Redding, Joseph D, Hugh L. MacNeil, and William C. Murdoch. 1893. Thirteenth Biennial Report of the State Board of Fish Commissioners. California: California. Dept. of Fish and Game,.
22 McPhail, J. D., and C. C. Lindsey. 1970. Freshwater fishes of northwestern Canada and Alaska. Fish Res Board Can 173: 1–381.
23 Major, R. L., J. Ito, S. Ito, and H. Godfrey. 1978. Distribution and origin of Chinook salmon (Oncorhynchus tshawytscha) in offshore waters of the North Pacific Ocean.
24 Carl, L. M. 1982. Natural Reproduction of Coho Salmon and Chinook Salmon in Some Michigan Streams. North American Journal of Fisheries Management 2: 375–380. https://doi.org/https://doi.org/10.1577/1548-8659(1982)2<375:NROCSA>2.0.CO;2.
25 Heifetz, Jonathan. 1982. Use Of Radio Telemetry To Study Upriver Migration Of Adult Κlaμath River Chinook Salmon. Arcata, California: Humboldt State University.
26 Rich, Willis H. 1942. The Salmon Runs of the Columbia River in 1938. Fish. Bull. Fish Wildl. Serv. 50: 101–147.
27 Rich, Willis Horton. 1925. Growth and Degree of Maturity of Chinook Salmon in the Ocean. U.S. Government Printing Office.
28 Stone, Livingstone. 1884. The quinnat salmon or California salmon—Oncorhynchus chouicha. The fisheries and fishery industries of the United States. Section I. Natural history of useful aquatic animals: 479–485.
29 Marrone, G. M., and D. A. Stout. 1997. 1997 Whitlocks Bay spawning station annual report. Annual Report 97–19. Pierre: South Dakota Department of Game, Fish and Parks.
30 Barnes, Michael E, Robert P Hanten, Rick J Cordes, William A Sayler, and John Carreiro. 2000. Reproductive Performance of Inland Fall Chinook Salmon. North American Journal of Aquaculture 62: 203–2011.
31 Biela, Vanessa R. von, Lizabeth Bowen, Stephen D. McCormick, Michael P. Carey, Daniel S. Donnelly, Shannon Waters, Amy M. Regish, et al. 2020. Evidence of prevalent heat stress in Yukon River Chinook salmon. Canadian Journal of Fisheries and Aquatic Sciences. 1840 Woodward Drive, Suite 1, Ottawa, ON K2C 0P7. https://doi.org/10.1139/cjfas-2020-0209.
32 Berejikian, B. A., E. P. Tezak, and A. L. LaRae. 2000. Female mate choice and spawning behaviour of Chinook salmon under experimental conditions. Journal of Fish Biology 57: 647–661. https://doi.org/https://doi.org/10.1111/j.1095-8649.2000.tb00266.x.
33 Gray, Robert H., and James M. Haynes. 1979. Spawning Migration of Adult Chinook Salmon ( Oncorhynchus tshawytscha ) Carrying External and Internal Radio Transmitters. Journal of the Fisheries Research Board of Canada 36: 1060–1064. https://doi.org/10.1139/f79-148.
34 Vronskiy, B. B. 1972. Reproductive biology of the Kamchatka River chinook salmon (Oncorhynchus tshawytscha (Walbaum)). Journal of Ichthyology 12: 259–273.
35 Allen, Mark A, and Thomas J Hassler. 1986. Species profiles: life histories and environmental requirements of coastal fishes and invertebrates (Pacific Southwest): Chinook salmon. Biological Report 82(11.49). U.S. Army Corps of Engineers, TR EL-82-4.: U.S. Fish and Wildlife Service. 1983-19.
36 Garner, S. R., B. N. Madison, N. J. Bernier, and B. D. Neff. 2008. Juvenile growth and aggression in diploid and triploid Chinook salmon Oncorhynchus tshawytscha (Walbaum). Journal of Fish Biology 73: 169–185. https://doi.org/10.1111/j.1095-8649.2008.01923.x.
37 Reimers, Paul E. 1968. Social Behavior among Juvenile Fall Chinook Salmon. Journal of Fisheries Research Board of Canada 25: 2005–2008.
38 Taylor, Eric B., and P. A. Larkin. 1986. Current Response and Agonistic Behavior in Newly Emerged Fry of Chinook Salmon, Oncorhynchus tshawytscha , from Ocean- and Stream- Type Populations. Canadian Journal of Fisheries and Aquatic Sciences 43: 565–573. https://doi.org/10.1139/f86-067.
39 Dill, Lawrenc M. 1969. The sub-gravel behaviour of Pacific salmon larvae. In Symposium on Salmon and Trout in Streams. HR MacMillan Lectures in Fisheries. Institute of Fisheries, University of British Columbia, Vancouver, 89–99.
40 Cogliati, Karen M., Crystal L. Herron, David L.G. Noakes, and Carl B. Schreck. 2019. Reduced stress response in juvenile Chinook Salmon reared with structure. Aquaculture 504: 96–101. https://doi.org/10.1016/j.aquaculture.2019.01.056.
41 Rosburg, Alex J, Brian L Fletcher, Michael E Barnes, Cody E Treft, and Blaise R Bursell. 2019. Vertically-Suspended Environmental Enrichment Structures Improve the Growth of Juvenile Landlocked Fall Chinook Salmon. International Journal of Innovative Studies in Aquatic Biology and Fisheries 5: 17–24.
42 Voorhees, Jill M., Nathan Huysman, Eric Krebs, and Michael E. Barnes. 2021. Exercise and Structure Improve Juvenile Chinook Salmon Rearing Performance. Open Journal of Marine Science 11: 80–91. https://doi.org/10.4236/ojms.2021.112006.
43 Sharpe, Cameron S, Daniel A Thompson, H Lee Blankenship, and Carl B Schreck. 1998. Effects of Routine Handling and Tagging Procedures on Physiological Stress Responses in Juvenile Chinook Salmon. The Progressive Fish-Culturist 60: 81–87.
44 Barton, Bruce A, Carl B Schreck, and Linda A Sigismondi. 1986. Multiple Acute Disturbances Evoke Cumulative Physiological Stress Responses in Juvenile Chinook Salmon. Transactions of the American Fisheries Society 115: 245–251.
45 De Clercq, A, M R Perrott, P S Davie, M A Preece, A Huysseune, and P E Witten. 2018. The external phenotype-skeleton link in post-hatch farmed Chinook salmon ( Oncorhynchus tshawytscha ). Journal of Fish Diseases 41: 511–527. https://doi.org/10.1111/jfd.12753.
46 Davie, Peter S., Seumas P. Walker, Matthew R. Perrott, Jane E. Symonds, Mark Preece, Adelbert De Clercq, and John S. Munday. 2018. Vertebral abnormalities in free-living Chinook salmon (Oncorhynchus tshawytscha, Walbaum) in New Zealand. New Zealand Journal of Marine and Freshwater Research 52: 444–456. https://doi.org/10.1080/00288330.2018.1455717.
47 Fletcher, G. C., G. Summers, V. Corrigan, S. Cumarasamy, and J. P. Dufour. 2002. Spoilage of King Salmon (Oncorhynchus tshawytscha) Fillets Stored Under Different Atmospheres. Journal of Food Science 67: 2362–2374. https://doi.org/https://doi.org/10.1111/j.1365-2621.2002.tb09555.x.
48 Robb, D H F, and S C Kestin. 2002. Methods Used to Kill Fish: Field Observations and Literature Reviewed. Animal Welfare 11: 269–282.
49 Lines, J. A., D. H. Robb, S. C. Kestin, S. C. Crook, and T. Benson. 2003. Electric stunning: a humane slaughter method for trout. Aquacultural Engineering 28: 141–154. https://doi.org/10.1016/S0144-8609(03)00021-9.
50 European Food Safety Authority (EFSA). 2009. Species-specific welfare aspects of the main systems of stunning and killing of farmed fish: Rainbow Trout. EFSA Journal 7: 1012. https://doi.org/10.2903/j.efsa.2009.1012.
51 Concollato, Anna, Rolf Erik Olsen, Sheyla Cristina Vargas, Antonio Bonelli, Marco Cullere, and Giuliana Parisi. 2016. Effects of stunning/slaughtering methods in rainbow trout (Oncorhynchus mykiss) from death until rigor mortis resolution. Aquaculture 464: 74–79. https://doi.org/10.1016/j.aquaculture.2016.06.009.
52 Humane Slaughter Association. 2018. Humane slaughter of finfish farmed around the world. Humane Slaughter Association.
53 Teletchea, Fabrice, and Pascal Fontaine. 2012. Levels of domestication in fish: implications for the sustainable future of aquaculture. Fish and Fisheries 15: 181–195. https://doi.org/10.1111/faf.12006.
54 Fowler, L. G. 1980. Substitution of Soybean and Cottonseed Products for Fish Meal in Diets Fed to Chinook and Coho Salmon. The Progressive Fish-Culturist 42: 87–91. https://doi.org/10.1577/1548-8659(1980)42[87:SOSACP]2.0.CO;2.
55 Higgs, David A., Jack R. McBride, Jack R. Markert, Bakhshish S. Dosanjh, M. Dianne Plotnikoff, and W. Craig Clarke. 1982. Evaluation of Tower and Candle rapeseed (canola) meal and Bronowski rapeseed protein concentrate as protein supplements in practical dry diets for juvenile chinook salmon (Oncorhynchus tshawytscha). Aquaculture 29: 1–31. https://doi.org/10.1016/0044-8486(82)90030-8.
56 Fowler, L. G. 1990. Feather meal as a dietary protein source during parr-smolt transformation in fall chinook salmon. Aquaculture 89: 301–314. https://doi.org/10.1016/0044-8486(90)90134-9.
57 Doughty, Katarina H., Shawn R. Garner, Mark A. Bernards, John W. Heath, and Bryan D. Neff. 2019. Effects of dietary fishmeal substitution with corn gluten meal and poultry meal on growth rate and flesh characteristics of Chinook salmon (Oncorhynchus tshawytscha). International Aquatic Research 11: 325–334. https://doi.org/10.1007/s40071-019-00241-3.
58 Grant, Amelia A. M., Daniel Baker, Dave A. Higgs, Colin J. Brauner, Jeffrey G. Richards, Shannon K. Balfry, and Patricia M. Schulte. 2008. Effects of dietary canola oil level on growth, fatty acid composition and osmoregulatory ability of juvenile fall chinook salmon (Oncorhynchus tshawytscha). Aquaculture 277: 303–312. https://doi.org/10.1016/j.aquaculture.2008.02.032.
59 Mugrditchian, Doris S., Ronald W. Hardy, and Wayne T. Iwaoka. 1981. Linseed oil and animal fat as alternative lipid sources in dry diets for chinook salmon (Oncorhynchus tshawytscha). Aquaculture 25: 161–172. https://doi.org/10.1016/0044-8486(81)90178-2.
60 Huang, S. S. Y., C. H. L. Fu, D. A. Higgs, S. K Balfry, P. M. Schulte, and C. J. Brauner. 2008. Effects of dietary canola oil level on growth performance, fatty acid composition and ionoregulatory development of spring chinook salmon parr, Oncorhynchus tshawytscha. Aquaculture 274: 109–117. https://doi.org/10.1016/j.aquaculture.2007.11.011.
61 Booman, Marije, Ian Forster, John C. Vederas, David B. Groman, and Simon R. M. Jones. 2018. Soybean meal-induced enteritis in Atlantic salmon (Salmo salar) and Chinook salmon (Oncorhynchus tshawytscha) but not in pink salmon (O. gorbuscha). Aquaculture 483: 238–243. https://doi.org/10.1016/j.aquaculture.2017.10.025.