Information
Version: B | 1.3 (2022-07-20)
WelfareScore | farm
Condensed assessment of the species' likelihood and potential for good fish welfare in aquaculture, based on ethological findings for 10 crucial criteria.
- Li = Likelihood that the individuals of the species experience good welfare under minimal farming conditions
- Po = Potential of the individuals of the species to experience good welfare under high-standard farming conditions
- Ce = Certainty of our findings in Likelihood and Potential
WelfareScore = Sum of criteria scoring "High" (max. 10)
General remarks
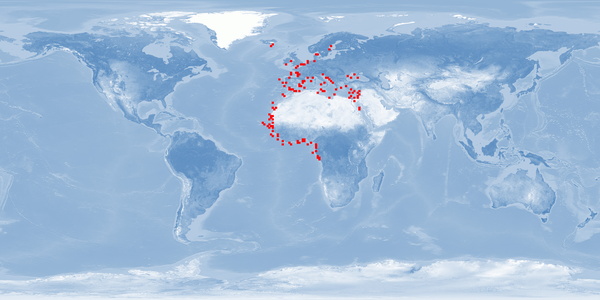
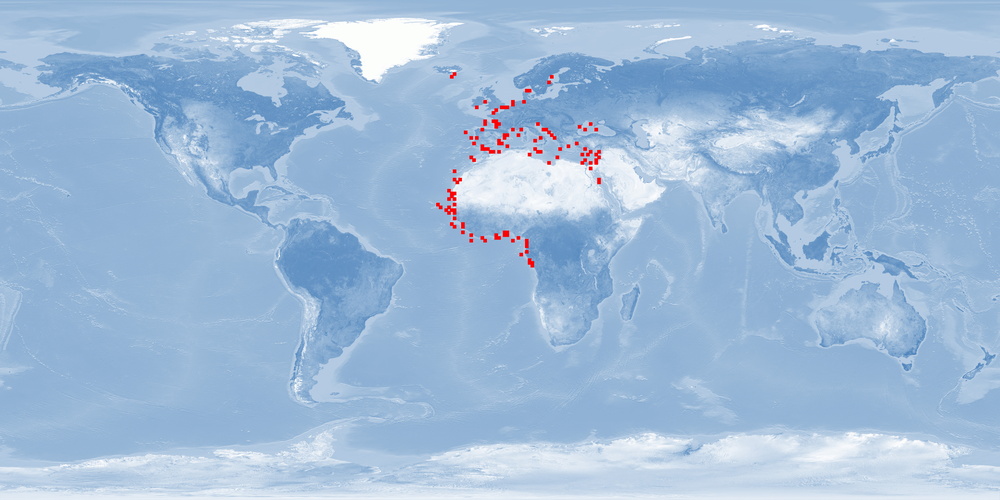
Agyrosomus regius is a migratory fish species which belongs to the Sciaenidae family. It is distributed in the Eastern Atlantic, from Norway to Gibraltar and Congo, and it is punctually present in some areas within the Mediterranean Sea. In the last decade, A. regius has become an increasingly important fish species to Mediterranean aquaculture. Several biological characteristics such as capability to withstand diverse environmental conditions, fast growth, and a good feed conversion rate when fed dry formulated feeds make A. regius an interesting candidate for aquaculture. Nevertheless, there are biological aspects such as the depth range and migration that would be difficult to respect in usual farming conditions. However, some of the present rearing conditions could be optimised to improve welfare. Stress responses to common farming conditions could be minimised using appropriate light conditions, densities, and diets according to the nutritional requirements of the species. Further research is needed on the development of a humane stunning and slaughter protocol, incidence of malformation, aggression, and substrate needs.
1 Home range
Many species traverse in a limited horizontal space (even if just for a certain period of time per year); the home range may be described as a species' understanding of its environment (i.e., its cognitive map) for the most important resources it needs access to.
What is the probability of providing the species' whole home range in captivity?
It is unclear for minimal and high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


2 Depth range
Given the availability of resources (food, shelter) or the need to avoid predators, species spend their time within a certain depth range.
What is the probability of providing the species' whole depth range in captivity?
It is low for minimal and high-standard farming conditions. Our conclusion is based on a high amount of evidence.


3 Migration
Some species undergo seasonal changes of environments for different purposes (feeding, spawning, etc.), and to move there, they migrate for more or less extensive distances.
What is the probability of providing farming conditions that are compatible with the migrating or habitat-changing behaviour of the species?
It is low for minimal and high-standard farming conditions. Our conclusion is based on a high amount of evidence.


4 Reproduction
A species reproduces at a certain age, season, and sex ratio and possibly involving courtship rituals.
What is the probability of the species reproducing naturally in captivity without manipulation of theses circumstances?
It is low for minimal farming conditions. It is high for high-standard farming conditions. Our conclusion is based on a high amount of evidence.


5 Aggregation
Species differ in the way they co-exist with conspecifics or other species from being solitary to aggregating unstructured, casually roaming in shoals or closely coordinating in schools of varying densities.
What is the probability of providing farming conditions that are compatible with the aggregation behaviour of the species?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


6 Aggression
There is a range of adverse reactions in species, spanning from being relatively indifferent towards others to defending valuable resources (e.g., food, territory, mates) to actively attacking opponents.
What is the probability of the species being non-aggressive and non-territorial in captivity?
It is unclear for minimal and high-standard farming conditions. Our conclusion is based on a low amount of evidence.


7 Substrate
Depending on where in the water column the species lives, it differs in interacting with or relying on various substrates for feeding or covering purposes (e.g., plants, rocks and stones, sand and mud, turbidity).
What is the probability of providing the species' substrate and shelter needs in captivity?
It is unclear for minimal and high-standard farming conditions. Our conclusion is based on a low amount of evidence.


8 Stress
Farming involves subjecting the species to diverse procedures (e.g., handling, air exposure, short-term confinement, short-term crowding, transport), sudden parameter changes or repeated disturbances (e.g., husbandry, size-grading).
What is the probability of the species not being stressed?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


9 Malformations
Deformities that – in contrast to diseases – are commonly irreversible may indicate sub-optimal rearing conditions (e.g., mechanical stress during hatching and rearing, environmental factors unless mentioned in crit. 3, aquatic pollutants, nutritional deficiencies) or a general incompatibility of the species with being farmed.
What is the probability of the species being malformed rarely?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


10 Slaughter
The cornerstone for a humane treatment is that slaughter a) immediately follows stunning (i.e., while the individual is unconscious), b) happens according to a clear and reproducible set of instructions verified under farming conditions, and c) avoids pain, suffering, and distress.
What is the probability of the species being slaughtered according to a humane slaughter protocol?
It is low for minimal farming conditions. It is medium for high-standard farming conditions. Our conclusion is based on a medium amount of evidence.


Side note: Domestication
Teletchea and Fontaine introduced 5 domestication levels illustrating how far species are from having their life cycle closed in captivity without wild input, how long they have been reared in captivity, and whether breeding programmes are in place.
What is the species’ domestication level?
DOMESTICATION LEVEL 4 27, level 5 being fully domesticated.
Side note: Forage fish in the feed
450-1,000 milliard wild-caught fishes end up being processed into fish meal and fish oil each year which contributes to overfishing and represents enormous suffering. There is a broad range of feeding types within species reared in captivity.
To what degree may fish meal and fish oil based on forage fish be replaced by non-forage fishery components (e.g., poultry blood meal) or sustainable sources (e.g., soybean cake)?
All age classes: WILD: carnivorous 28 17. FARM: fish meal and fish oil may be partly* replaced by non-forage fishery components 29 30 31.
*partly = <51% – mostly = 51-99% – completely = 100%
Glossary
BENTHOPELAGIC = living and feeding near the bottom of a body of water, floating above the floor
DOMESTICATION LEVEL 4 = entire life cycle closed in captivity without wild inputs 27
FARM = setting in farming environment or under conditions simulating farming environment in terms of size of facility or number of individuals
IND = individuals
JUVENILES = fully developed but immature individuals, for details ➝ Findings 10.1 Ontogenetic development
LAB = setting in laboratory environment
LARVAE = hatching to mouth opening, for details ➝ Findings 10.1 Ontogenetic development
OCEANODROMOUS = living and migrating in the sea
SPAWNERS = adults during the spawning season; in farms: adults that are kept as broodstock
WILD = setting in the wild
Bibliography
2 Barata, Marisa. 2015. Pré-engorda e engorda de corvina. Master, Peniche: Escola Superior de Turismo e Tecnologia do Mar- Instituto Politecnico de Leiria.
3 Stipa, P., and M. Angelini. 2005. Cultured Aquatic Species Information Programme. Argyrosomus regius. Rome: FAO Fisheries and Aquaculture Department.
4 Duncan, N. J., A. Estévez, H. Fernández-Palacios, I. Gairin, C. M. Hernández-Cruz, J. Roo, D. Schuchardt, and R. Vallés. 2013. Advances in aquaculture hatchery technology: 17. Aquaculture production of meagre. Woodhead Publishing.
5 Griffiths, M. H., and P. C. Heemstra. 1995. A contribution to the taxonomy of the marine fish genus Argyrosomus (Perciformes: Sciaenidae), with descriptions of two new species from southern Africa. Ichtyological Bulletin 65.
6 Shabana, N. M. A., S. H. Abd El Rhaman, M. A. Al Absawy, and S. S. Assem. 2012. Reproductive biology of Argyrosomos regius (Asso, 1801) inhabiting the south eastern Mediterranean Sea, Egypt. Egyptian Journal of Aquatic Research 38: 147–156.
7 González-Quirós, Rafael, Juan del Árbol, María del Mar García-Pacheco, Alfonso J. Silva-García, José María Naranjo, and Beatriz Morales-Nin. 2011. Life-history of the meagre Argyrosomus regius in the Gulf of Cádiz (SW Iberian Peninsula). Fisheries Research 109: 140–149. https://doi.org/10.1016/j.fishres.2011.01.031.
8 Morales-Nin, B., A. J. Geffen, S. Pérez-Mayol, M. Palmer, R. González-Quirós, and A. Grau. 2012. Seasonal and ontogenic migrations of meagre (Argyrosomus regius) determined by otolith geochemical signatures. Fisheries Research 127: 154–165. https://doi.org/10.1016/j.fishres.2012.02.012.
9 Vallés, Roser, and Alicia Estévez. 2013. Light conditions for larval rearing of meagre (Argyrosomus regius). Aquaculture 376: 15–19. https://doi.org/10.1016/j.aquaculture.2012.11.011.
10 Duncan, Neil, Alicia Estévez, Javier Porta, Ignacio Carazo, Fernando Norambuena, Cristobal Aguilera, Ignaci Gairin, Francesco Bucci, Roser Valles, and Constantinos C. Mylonas. 2012. Reproductive development, GnRHa-induced spawning and egg quality of wild meagre (Argyrosomus regius) acclimatised to captivity. Fish Physiology and Biochemistry 38: 1273–1286. https://doi.org/10.1007/s10695-012-9615-3.
11 Mylonas, Constantinos C., Nikolaos Mitrizakis, Maria Papadaki, and Irini Sigelaki. 2013. Reproduction of hatchery-produced meagre Argyrosomus regius in captivity I. Description of the annual reproductive cycle. Aquaculture 414: 309–317. https://doi.org/10.1016/j.aquaculture.2013.09.009.
12 Estévez, A., L. Treviño, and E. Gisbert. 2007. La densidad larvaria inicial afecta al crecimiento pero no a la supervivencia de las larvas de corvina (Argyrosomus regius) en cultivo. In , 747–750. Vigo, Spain: Cerviño, A., Guerra, A., Pérez, C. (Eds.).
13 Quéro, J.C. 1989. Sur la piste des maigres Argyrosomus regius (Pisces, Sciaenidae) du Golfe de Gascogne et de Mauritanie. Océanis 15: 161–170.
14 Quéméner, L. 2002. Le maigre commun (Argyrosomus regius). Biologie, peche, marche et potentiel aquacole. France: INFREMER.
15 Catalán, I. A., M. T. Jiménez, J. I. Alconchel, L. Prieto, and J. L. Muñoz. 2006. Spatial and temporal changes of coastal demersal assemblages in the Gulf of Cadiz (SW Spain) in relation to environmental conditions. Deep Sea Research Part II: Topical Studies in Oceanography 53. The Gulf of Cadiz Oceanography: A Multidisciplinary View: 1402–1419. https://doi.org/10.1016/j.dsr2.2006.04.005.
16 Millán-Cubillo, Antonio Francisco, Juan Antonio Martos-Sitcha, Ignacio Ruiz-Jarabo, Salvador Cárdenas, and Juan Miguel Mancera. 2016. Low stocking density negatively affects growth, metabolism and stress pathways in juvenile specimens of meagre (Argyrosomus regius, Asso 1801). Aquaculture 451: 87–92. https://doi.org/10.1016/j.aquaculture.2015.08.034.
17 Jiménez, M. T., E. Pastor, A. Grau, J. I. Alconchel, R. Sánchez, and S. Cárdenas. 2005. Revisión del cultivo de esciénidos en el mundo, con especial atención a la corvina Argyrosomus regius (Asso, 1801). Bol. Inst. Esp. Oceanogr. 21: 169–175.
18 Oviedo, María del Mar G. 2013. Recovery of meagre (Argyrosomus regius) population in the Balearic coastal ecosystem (Western Mediterranean). Ph.D. dissertation, University of the Balearic Islands.
19 Campoverde, Cindy, Covadonga Rodriguez, José Perez, Enric Gisbert, and Alicia Estévez. 2017. Early weaning in meagre Argyrosomus regius: Effects on growth, survival, digestion and skeletal deformities. Aquaculture Research 48: 5289–5299. https://doi.org/10.1111/are.13342.
20 Diken, Gurkan, Orhan Demir, and Mehmet Naz. 2016. The potential effects of commercial feeding protocol on protease activities and cortisol stress responses of meagre (Argyrosomus regius). Agriculture and Food.
21 Samaras, Athanasios, Nikos Papandroulakis, Marilena Costari, and Michail Pavlidis. 2016. Stress and metabolic indicators in a relatively high (European sea bass, Dicentrarchus labrax) and a low (meagre, Argyrosomus regius) cortisol responsive species, in different water temperatures. Aquaculture Research 47: 3501–3515. https://doi.org/10.1111/are.12800.
22 Cardeira, J., R. Vallés, G. Dionísio, A. Estévez, E. Gisbert, P. Pousão-Ferreira, M. L. Cancela, and P. J. Gavaia. 2012. Osteology of the axial and appendicular skeletons of the meagre Argyrosomus regius (Sciaenidae) and early skeletal development at two rearing facilities. Journal of Applied Ichthyology 28: 464–470. https://doi.org/10.1111/j.1439-0426.2012.01979.x.
23 Soares, F., I. Fernández, B. Costas, and P. J. Gavaia. 2014. Non-infectious disorders of warmwater fish. In Diseases and Disorders of Finfish in Cage Culture, 2nd ed. CAB International.
24 European Food Safety Authority (EFSA). 2008. Scientific opinion of the panel on animal health and welfare on a request from the european commission on animal welfare aspects of husbandry systems for farmed european seabass and gilthead seabream. EFSA Journal 844: 1–21.
25 Cárdenas, S. 2010. Crianza de la corvina (Argyrosomus regius). 3. Cuadernos de Acuicultura.
26 European Food Safety Authority (EFSA). 2009. Species-specific welfare aspects of the main systems of stunning and killing of farmed Seabass and Seabream. EFSA Journal 1010: 1–52. https://doi.org/10.2903/j.efsa.2009.1010.
27 Teletchea, Fabrice, and Pascal Fontaine. 2012. Levels of domestication in fish: implications for the sustainable future of aquaculture. Fish and Fisheries 15: 181–195. https://doi.org/10.1111/faf.12006.
28 Cabral, E. M., and Barbara Ohmert. 2001. Diet of juvenile meagre, Argyrosomos regius, within the Tagus estuary. Cahiers de biologie marine 42: 289–293.
29 Barroso, Carolina. 2014. Effects of partial replacement of fish meal by carob seed germ meal on growth performance and immune parameters of meagre (Argyrosomus regius, Asso, 1801). Master, Porto: Faculdade de Ciências da Universidade do Porto.
30 Emre, Yılmaz, Adem Kurtoğlu, Nesrin Emre, Betül Güroy, and Derya Güroy. 2016. Effect of replacing dietary fish oil with soybean oil on growth performance, fatty acid composition and haematological parameters of juvenile meagre, Argyrosomus regius. Aquaculture Research 47: 2256–2265. https://doi.org/10.1111/are.12677.
31 Ribeiro, Laura, João Moura, Marta Santos, Rita Colen, Vera Rodrigues, Narcisa Bandarra, Florbela Soares, et al. 2015. Effect of vegetable based diets on growth, intestinal morphology, activity of intestinal enzymes and haematological stress indicators in meagre (Argyrosomus regius). Aquaculture 447. Research for the Next 40 Years of Sustainable Global Aquaculture: 116–128. https://doi.org/10.1016/j.aquaculture.2014.12.017.